 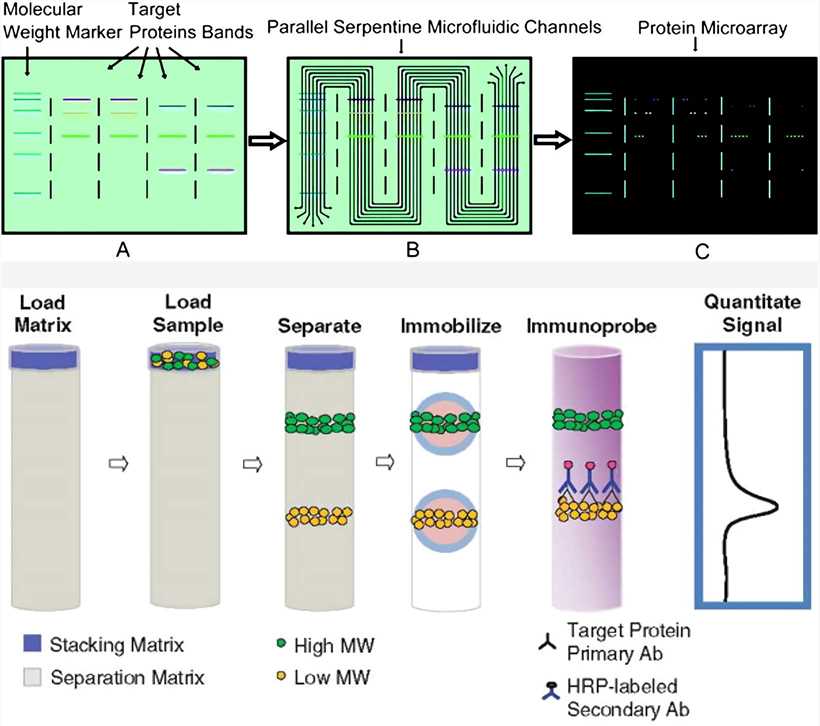
Intuitively, this is imperative for a valid experiment as unequal proteins per lane can skew the analysis. There must be an equal concentration of proteins per western blot sample. Collectively, this information underscores the need to tailor protein extraction to sample type and the target protein. PPIs are used to maintain the structure and phosphorylation status of the target protein from the activity of endogenous phosphatases upon cell lysis and exogenous phosphatases in the lysis microenvironment. As such, gentle buffers without detergents are required. Although rare, some antibodies will not be able to detect denatured samples. For example, radioimmunoprecipitation assay buffer (RIPA) is more adept for nuclear and mitochondrial proteins. Furthermore, the cell lysis buffer used in extraction should align with target protein cellular localization. For example, most tissue preparation is by homogenization or sonication however, osmotic shock or detergent lysis is more suited for easily lysed cells such as erythrocytes or cultured cells. There are numerous methods of extraction, and proper selection depends on the sample type. Western blot samples are first prepared by protein extraction with specialized cell lysis buffers and protease and phosphatase inhibitors (PPIs). Proper sample preparation for subsequent electrophoresis is crucial for downstream analysis. Western blot relies on the principles of equal loading of proteins, separation of proteins by molecular weight, electrophoretic transfer to a suitable membrane, and probing of antibodies. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
